The level of adherence to PrEP appears to be the key variable in achieving effective prevention, writes Dr David Baker
GPs have a new tool in the prevention of HIV with the listing of pre-exposure prophylaxis (PrEP) on the Pharmaceutical Benefits Scheme (PBS). This means that all GPs can prescribe PrEP with dispensing from community pharmacies.
The most widely used form of PrEP is a once-daily tablet containing two antiretrovirals – tenofovir and emtricitabine (TDF/FTC, brand name Truvada) – which is the medication listed on the PBS. Unlike post-exposure prophylaxis (PEP), which is taken after a potential exposure to HIV, PrEP is taken on an ongoing basis before potentially being exposed to HIV.
PrEP involves people at risk of HIV taking antiretroviral medication to prevent infection, an approach which has proved to be highly effective in preventing HIV transmission.1 More than 9000 people in New South Wales are currently receiving PrEP through the EPIC trial with similar initiatives in other states.2
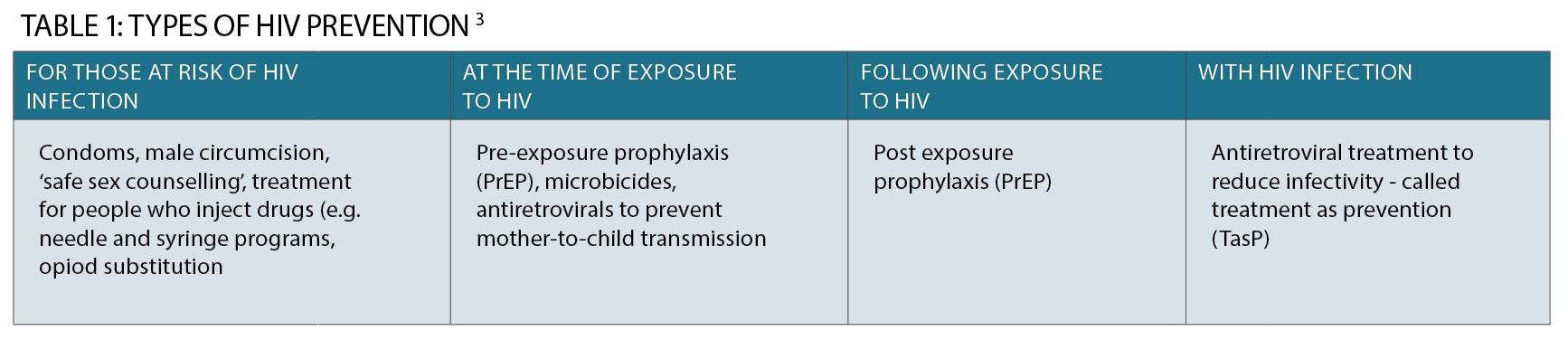
It is important to note that PrEP is a newer way to prevent HIV infection along with existing preventive strategies, such as condom use and the provision of clean injecting equipment.
Why do we need it?
The number of new HIV diagnoses in Australia has remained steady over the last five years at just over 1000 a year nationwide.4
Male-to-male sex continues to be the major HIV risk exposure and is associated with 70% of HIV diagnoses with 21% from heterosexual transmission.
This highlights the ongoing need to discuss risk factors, such as sexual behaviour, with all new patients. At risk patients then need to be offered HIV testing and prevention. An estimated 11% of people living with HIV are undiagnosed.4
Programs to “End HIV by 2020” focus on a variety of interventions, including frequent testing, early treatment and prevention activities.
Encouragingly, some states are seeing a decline in new HIV diagnoses, probably related to the scaling up of preventative activities, including the use of PrEP as well as earlier diagnosis and treatment.5
How effective is it?
The most widely studied form of PrEP is daily use of two anti-retroviral medications tenofovir with emtricitabine by HIV negative people to prevent infection with HIV. Many placebo-controlled randomised studies have shown a reduction in HIV transmission when PrEP is taken regularly.
Among men who have sex with men, the iPREX study reported a reduction of 44% in HIV incidence in the intention-to-treat analysis, however adherence was relatively poor.6
In the open label extension of iPREX however, no infections occurred among participants with dried blood spot levels of tenofovir consistent with taking at least four TDF/FTC pills per week.7
Among heterosexually active men and women, PrEP reduced the risk of getting HIV by 62%.
Participants who became infected had far less drug in their blood, compared with matched participants who remained uninfected.8 Significantly reduced risk has also been demonstrated in people who inject drugs.9
In all populations studied, a high level of adherence appears to be the key variable in achieving effective HIV prevention.
What are the potential side effects?
Most common side effects are mild and include headache, nausea, abdominal pain and weight loss.
These usually subside after a few weeks. Very rarely patients will experience severe side effects when starting PrEP and should be advised to present for urgent review to exclude lactic acidosis and severe hepatomegaly with steatosis.10
Of more concern are potential long-term side effects.
Truvada product information advises that TDT/FTC should not be used with creatinine clearance below 60 mL/min.11
Renal impairment, including acute renal failure and Fanconi syndrome (renal tubular injury with hypophosphatemia) have been reported.
Renal function should be monitored at baseline and every three to six months during PrEP therapy.1
A reduction in bone mineral density is associated with tenofovir treatment for HIV. Currently it is not known if this condition will be a long-term concern with the use of PrEP.
Tenofovir and emtricitabine are active against hepatitis B.
It is important to assess the need for hepatitis B treatment in patient with chronic hepatitis B infection.
Severe acute exacerbations of hepatitis B have been reported in patients who are co-infected with HBV and HIV and have discontinued TDF/FTC.
How to prescribe
Prescribing preventative medication is something that all GPs are familiar with, for example contraceptives or malaria chemoprophylaxis. As in these clinical situations, patient counselling, medication choice and monitoring can be quite complex. With the arrival of any new therapy additional training is advisable.
A list of online resources, as well as face-to-face training, is included at the end of this article.
Patients present in a variety of clinical situations: some may have heard about PrEP and may request treatment, others may have risk factors for HIV that you have identified, and you may then suggest PrEP as a possible HIV preventative option. Assessment for PrEP is also a great opportunity to review patients potential need for STI and hepatitis screening.
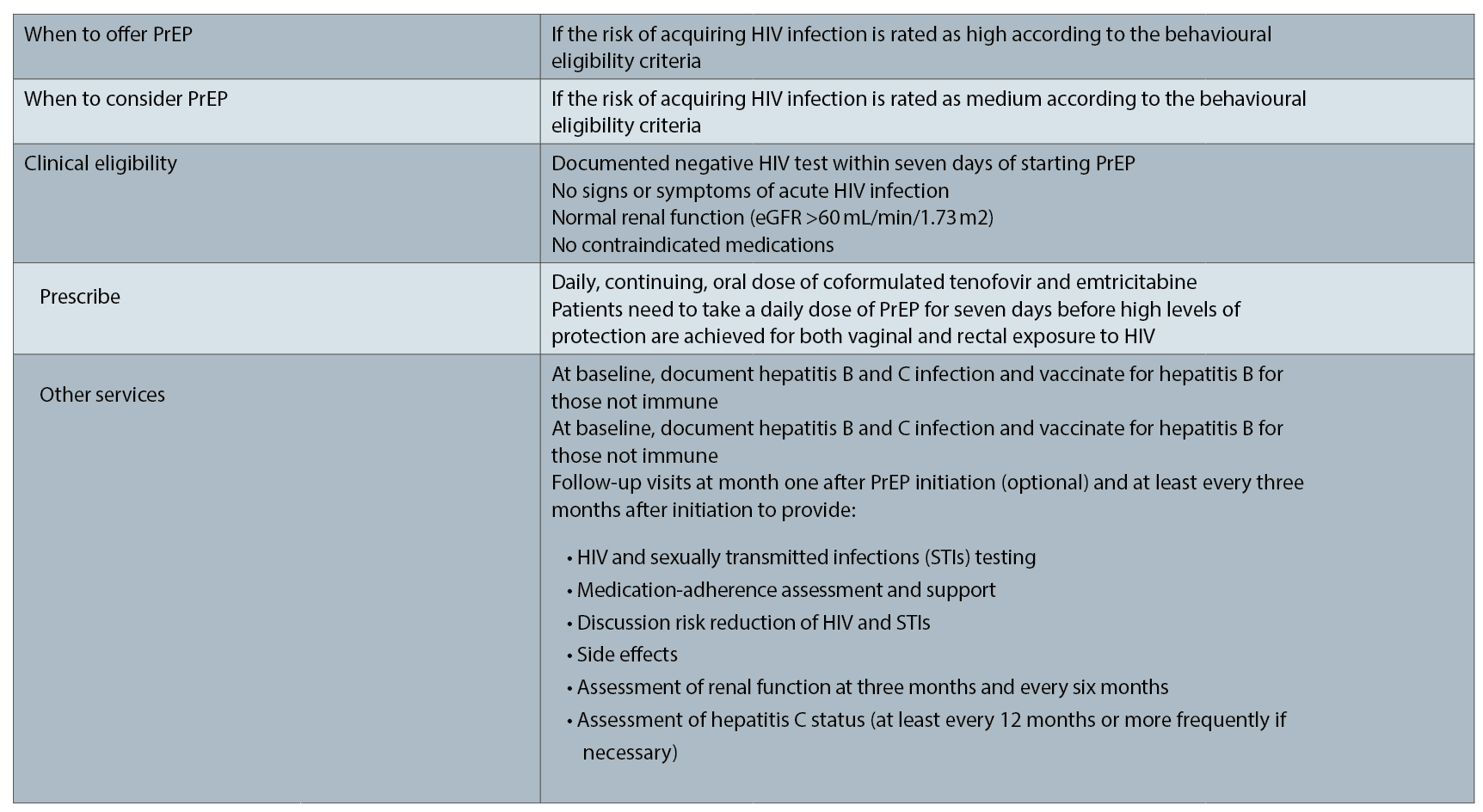
Behavioural eligibility
The steps in PrEP prescribing is based on the Australian guidelines.1 The first stage in prescribing PrEP is determining behavioural eligibility. This step can be quite demanding as you, the prescribing doctor, need to perform a risk assessment regarding the patient’s need for PrEP. This involves taking a sexual and drug use history.
The PBS criteria require that “treatment must be for patients at medium to high risk of HIV infection, as defined by the Australasian Society for HIV, Viral Hepatitis and Sexual Health Medicine (ASHM) Guidelines”. Essentially this requires consideration of behaviour (sexual such as vaginal or anal sex with or without condoms, injecting drug use) and the HIV status of the partner(s) if known or the prevalence of HIV in the contact population. Other markers for risk are also considered including amphetamine use or recent sexually transmitted infection.
ASHM have produced a two-page guide to assist in this assessment at http://www.ashm.org.au/products/product/3000100092
Taking long-term medication is a big step for most people. It is useful during this consultation to provide people with detailed written information about PrEP.
Clinical eligibility
Following a behavioural assessment, it is necessary to then perform a clinical assessment. This includes a baseline HIV, STI and hepatitis screen along with a check of renal function. Tenofovir, a component of PrEP, is renally excreted and is potentially nephrotoxic so it should not be used in the presence of renal disease or with an eGFR of less than 60. It should also not be prescribed with nephrotoxic medications (such as high dose nonsteroidal inflammatory drugs).
As with any treatment it is important to discuss potential short-term side effects and long-term side effects. Patients should be reviewed at one month then every three months.
Supply of PrEP
The final step in providing PrEP is organising the supply. Over the last two years PrEP has been available via various state-based trial and access programs with direct dispensing to patients from clinics.
From 1 April this year PrEP TDF/FTC has been listed as a streamlined authority (see table two) with dispensing from a community pharmacy at a cost of $39.50 per month or $6.40 per month with a concession card. Patients who aren’t eligible for PrEP or who don’t have a Medicare card can access TDF/FTC PrEP by:
• Trial participation – now winding up since PBS listing (but future trials may be planned)
• Private prescription with dispensing from community pharmacy (cost is about $250 a month)
• Compassionate access programs – with medication provided by pharmaceutical companies
• Direct importation from online suppliers via the TGA Personal Importation Scheme – see http://pan.org.au/ for details and pricing (as low as about $30 a month)
Note that the field off PrEP is rapidly evolving. Episodic “event driven” use of PrEP appears to be very effective and this approach is likely to be offered soon.14
Long-acting injectable forms of PrEP are in development and are looking very promising. They may be available via clinical trials in the future.

Summary
• The listing of PrEP on the PBS provides general practitioners with a vital new tool in the fight to prevent HIV.
• Providing PrEP also offers GPs a great opportunity to engage patients in regular health screening for other health problems, including STIs and hepatitis.
• Online training and guidelines for PrEP are available from ASHM (the Australasian Society for HIV, Viral Hepatitis and Sexual Health Medicine) and other training organisations.
Dr David Baker is a GP in Darlinghurst. He also works as a medical advisor to AHSM.
Disclosure: Dr David Baker has received payments from Gilead, ViiV and MSD for clinical trials, advisory board and travel grants
References:
1. Edwina Wright et al, Australasian Society for HIV, Viral Hepatitis and Sexual Health Medicine HIV pre-exposure prophylaxis: clinical guidelines, Journal of Viral Eradication, July 2017
2. NSW HIV Strategy 2016 – 2020 Quarter 4 & Annual 201Data Report http://www.health.nsw.gov.au/endinghiv/Pages/tools-and-data.aspx
3. Baker, D, Owen, L, General practitioners and HIV, http://hivmanagement.ashm.org.au/index.php/populations-and-situations/gps-and-hiv#
4. Kirby Institute. HIV, viral hepatitis and sexually transmissible infections in Australia: annual surveillance report 2017. Sydney: Kirby Institute, UNSW Sydney; 2017
5. Christine Selvey. Sponsored Satellite Session NSW Ministry of Health and the Kirby Institute: On our way to Ending HIV in NSW. Is this the end of the beginning? Australasian HIV and AIDS Conference, Canberra, 2017
6. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. The New England journal of medicine. 2010;363(27):2587-405 2599.
7. Grant RM, Anderson PL, McMahan V, et al. Uptake of pre-exposure prophylaxis, sexual practices, and HIV incidence in men and transgender women who have sex with men: a cohort study. Lancet Infect Dis. 2014;14(9):820-829.
8. Thigpen, M.C., et al., Antiretroviral preexposure prophylaxis for heterosexual HIV transmission in Botswana. N Engl J Med, 2012. 367(5): p. 423-34.
9. Choopanya, K., et al., Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet, 2013. 381(9883): p. 2083-90.
10. TRUVADA® Product Monograph www.gilead.ca
11. TRUVADA® Product Monograph www.gilead.ca
12. https://www.pbs.gov.au/medicine/item
13. http://www.ashm.org.au/products/product/3000100092
14. Antoni G et al. On-demand PrEP with TDF/FTC remains highly effective among MSM with infrequent sexual intercourse: a sub-study of the ANRS IPERGAY trial. IAS 2017, 23–26 July 2017, Paris. Oral abstract TUAC0102.