GPs can use the SmartVax data to reassure parents about the safety of the flu vaccine and of multiple concomitant vaccinations
A Perth GP has been awarded the AMA Excellence in Healthcare Award for renewing confidence in childhood vaccines through his innovative adverse events tracking system.
So far this year, Dr Alan Leeb’s SmartVax technology has followed up with around 105,500 patients who had received the influenza vaccine.
The technology works by automatically texting patients three days after a flu shot to ask whether they experienced any adverse events.
The best way to flag a potential problem with a vaccine was to monitor the rate of medical attendance, Dr Leeb (pictured) told The Medical Republic.
“In other words, did you attend a GP, after-hours clinic or emergency department in relation to that vaccination?” he said.
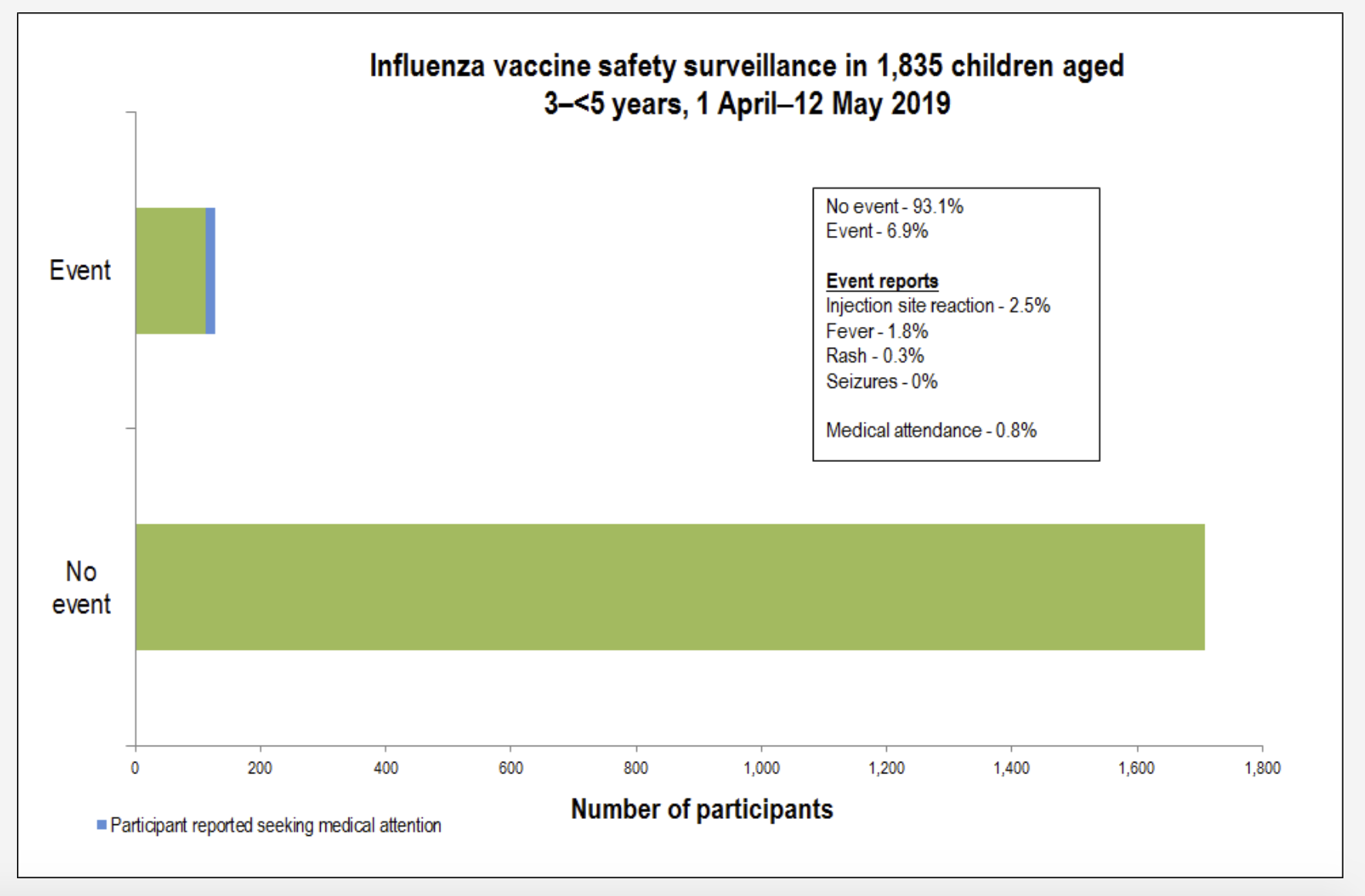
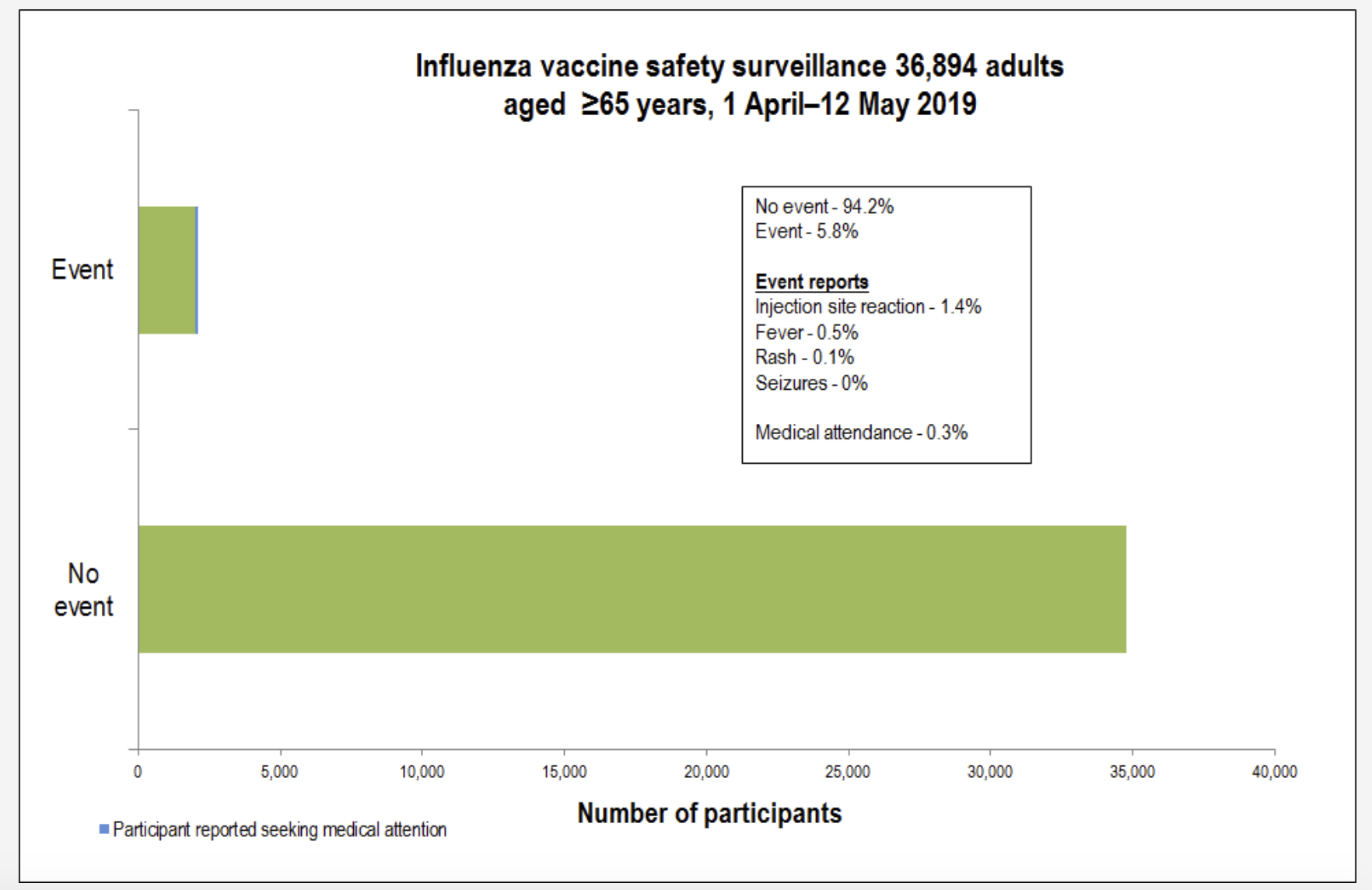
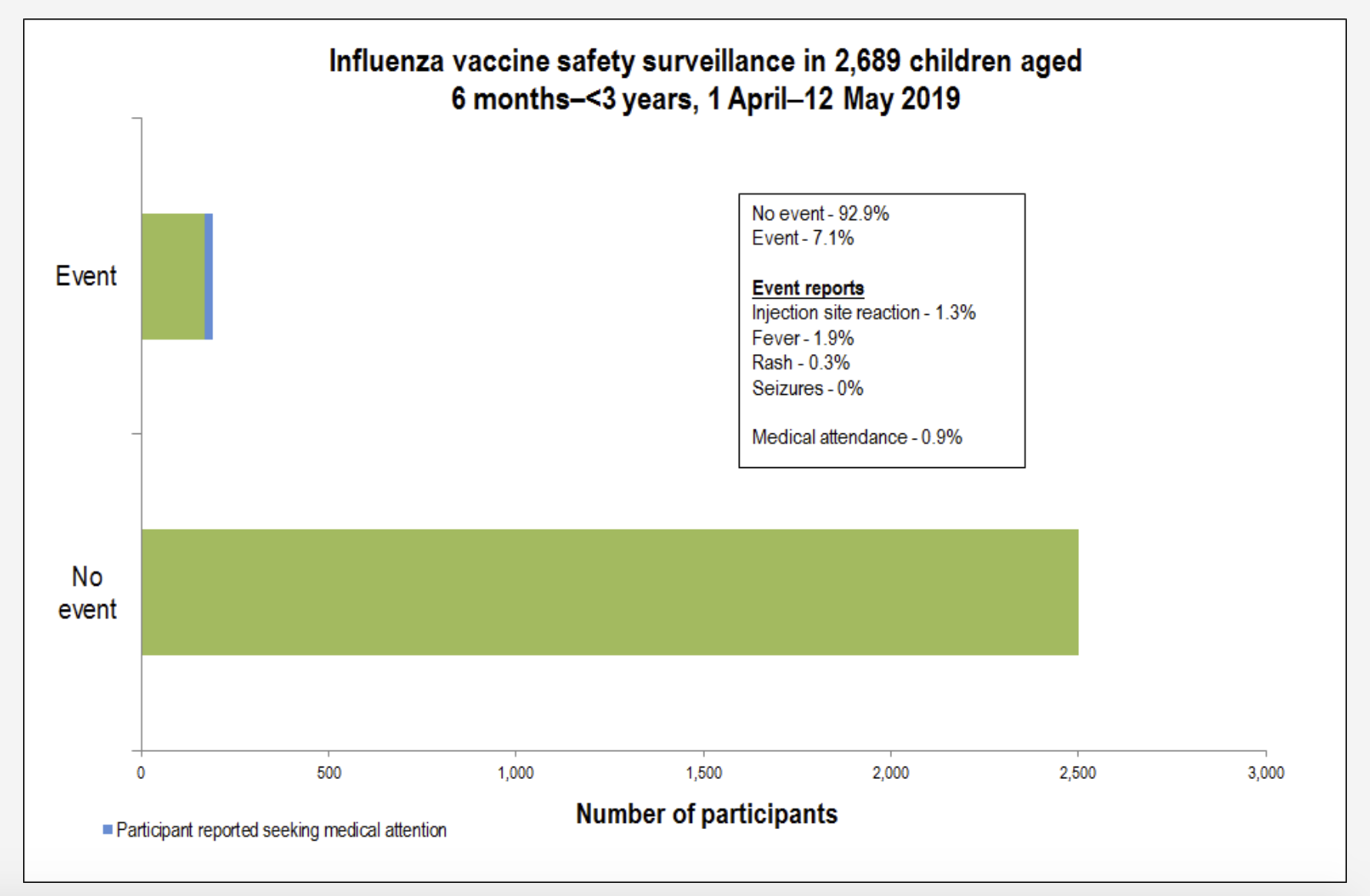
His data shows that the rate of medical attendance following the flu vaccination is 0.3% on average across all age groups.
“We’ve monitored flu vaccines since 2014 and we know that that vaccine is completely safe,” Dr Leeb said.
“We are not just saying that, we’ve got the data.
“Safe means we are not picking up any safety signals. So, we are getting a background reactivity rate of around 6% and its mainly minor local reactions or low-grade temperature, so very minor symptoms.
“We’ve had no reported serious events from flu vaccinations.”
The last time there was a spike in serious reactions to the influenza vaccine was in 2010, when around one-third of vaccinated children had an adverse event, and many wound up in hospital with febrile convulsions. Western Australia was the first state to offer free flu vaccines to children under the age of five and had high rates of uptake pre-2010.
“Our immunisation rates fell from 45% to 7% in 2011,” Dr Leeb said. “It just dropped like a stone. And probably until this year, our rates have never recovered. So, we’ve been running around the 10% rate since then.”
“The biggest issue was that prior to 2010, Australia had a passive system of adverse events surveillance,” he said.
“So, in other words, the only way you would know if there was a reaction to a vaccine was if each patient reported it manually to the TGA or local health authority.
“The beauty of SmartVax is that it is active surveillance so all immunised patients are contacted by SMS.
Around 75% of patients responded to the SMS, “which is huge”, he said.
The data is analysed by AusVaxSafety for reporting to government and public health authorities. Summarised data is available to the public through www.ausvaxsafety.org.au. GPs can access their local data via the SmatVax tool.
GPs could use the data to reassure parents about the safety of multiple concomitant vaccinations, he said.
“Currently, 12-month and 18-month old infants receive three vaccinations at each sitting. If you add flu vaccination, this would mean four vaccinations, two in the arm and two in the legs. That could be perceived as quite an onslaught for an already anxious parent.”
“But we know that concomitant vaccination, which is more than one vaccine at a time, is safe with no increased or additive effect on reactivity.
“It’s reassuring for parents when you can say, ‘This is local data. This is Australian data. We are not pulling data from the elsewhere’.”
The SmartVax data was particularly important for communicating the expected adverse event rates for highly-reactive vaccines, such as Bexsero.
“About 27% of children that get vaccinated with Bexsero will have a reaction,” Dr Leeb said.
“So, you can sit down with a parent and say, ‘Look we know that at least one in four of these kids is going to have a reaction from this vaccine, these are the reactions we expect and these are the reactions that would be of concern. So far, we have not detected any serious adverse events.”
Dr Leeb has also launched a tool called SmartStartAllergy to track the rates and effects of introducing allergenic foods within the first year of life.
The tool, which was developed in partnership with the National Allergy Strategy and Perth Children’s Hospital Foundation, is now running at 50 sites.
Reproduced with permission from SmartVax


