Higher transmission among the young could be due to the variant or vaccination patterns.
The Delta variant is surging across the globe, and the World Health Organization warns it will rapidly become the world’s dominant strain of COVID-19.
Delta is more infectious than the Alpha variant, and preliminary data suggest children and adolescents are at greater risk of becoming infected with this variant, and transmitting it.
Is this true? And with Sydney school students set to begin term 3 remotely, what’s the best way to manage school outbreaks?
Let’s take a look at the evidence.
Delta in children and young people
In the United Kingdom, where the Delta variant has been predominating since May, infections are rising fastest among 17-29-year-olds, who are mostly unvaccinated. Infections are also increasing in younger age groups, but at a lower rate.
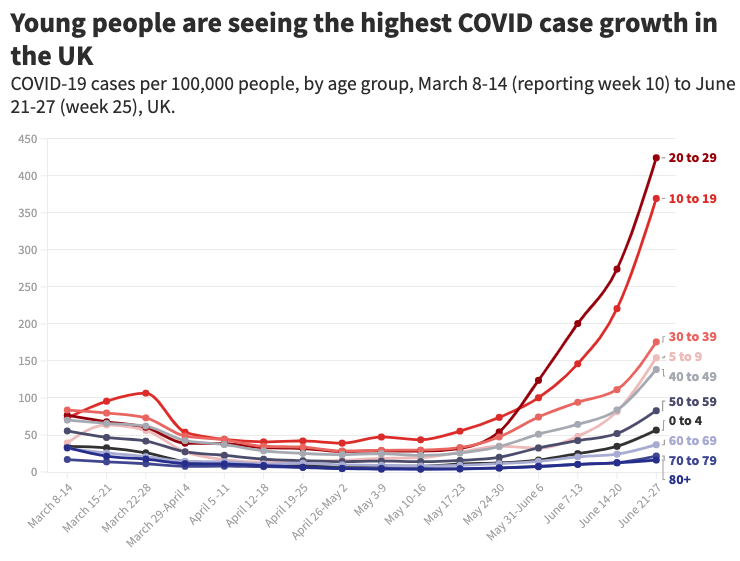
Overall, increased transmission among children and young people may partly be due to Delta. But also, in countries like the UK, these age groups are most susceptible to infection because older groups have been largely vaccinated.
While we don’t yet have data on the severity of illness in children associated with the Delta variant specifically, we know with COVID generally, kids are much less likely to become very unwell.
Research from the Murdoch Children’s Research Institute found children clear the virus more quickly than adults, which might go some way to explaining this.
How is Delta affecting transmission in schools?
In 2020, face-to-face learning wasn’t a significant contributor to community transmission in Victoria. Similarly, during the first wave in New South Wales, transmission rates were low in education settings. Concerns children may bring infections home to vulnerable family members weren’t supported by the evidence.
However, the situation is looking somewhat different now with the emergence of new variants and varying levels of vaccine coverage in different countries.
There does appear to be more transmission in schools. In the week ending June 27 there were outbreaks in 11 nursery schools, 78 primary schools, 112 secondary schools and 18 special needs schools in the UK.
While outbreaks in schools are increasing, the vast majority of transmission still occurs in households.
In 2021 in Australia, there have been very few school infections with Delta. In Western Australia, where schools have remained open, an infectious case attended three schools but this didn’t result in any school outbreaks.
During the current NSW outbreak, there have been several schools and early childhood centres with COVID-19 cases, and we have seen one outbreak at a primary school.
Although schools in Australia have largely been spared, transmission rates have been higher than we’ve seen with other variants. Almost all household contacts of cases are becoming infected.
In the recent Melbourne primary school outbreak, our research yet to be published showed that 100% of the household contacts of children who were infected at school went on to test positive.
Fortunately, testing, tracing and isolating were very effective in containing the outbreak, even with the Delta variant.
But these recent school outbreaks highlight why it’s so important adults of all ages, especially parents and teachers, get vaccinated.
Should we vaccinate children?
There are benefits of vaccinating children, particularly teenagers. These include direct protection against the disease, but also reducing transmission to vulnerable adults and enabling continued school attendance.
The risks and benefits need to be carefully calculated in a low transmission setting like Australia. In terms of risks, emerging data suggest the mRNA vaccines Pfizer and Moderna are associated with a very small risk of myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the heart lining) in young adolescents and adults, particularly males. Although most cases are mild, it can be a serious condition and is being closely monitored.
The United States, Canada, and a few countries in Europe are already vaccinating children over 12. Australia’s drug regulator is currently weighing this up.
For now, we should continue to vaccinate adults in priority groups. We have a long way to go to get the most vulnerable vaccinated first, and are still constrained by vaccine supply.
As we grapple with the benefits and risks for teenagers, it’s also worth asking them if they want to be vaccinated and why. Many have been adversely impacted by the pandemic and are desperate to move on with their lives.
What should parents look out for?
With the Delta variant, a headache, sore throat and runny nose are now the most commonly reported symptoms among unvaccinated people.
These symptoms have eclipsed fever and cough, the most common symptoms earlier in the pandemic.
So it’s imperative parents still take their children to be tested if they become unwell, even if the symptoms appear more like the common cold.
Where to from here?
When adults are more widely vaccinated and our borders open, school outbreaks will likely continue to happen. Even in places like Israel, where a high proportion of the population has received two doses, school outbreaks have recently occurred.
Australia needs a clear plan that outlines how best to keep schools open, while preventing transmission and keeping children and teachers safe during any outbreaks.
The Royal Australasian College of Physicians last week called for a national plan to this end.
This should include school staff being prioritised for vaccination.
And until we have high vaccination coverage, there’s evidence that well implemented school-based mitigation measures work to prevent transmission in education settings.
This could include a range of measures, adjusted according to risk, such as keeping non-essential adults off school grounds, mask use in high school students (and possibly primary students too), staggering timetables, reducing class sizes and improving classroom ventilation.
By monitoring the effects of new variants on children’s health, coupled with detailed risk-benefit analyses, we will determine the best time for children and adolescents to be vaccinated.
In the meantime, parents and all eligible adults can do their bit to protect children and reduce the risk of school outbreaks by getting vaccinated themselves.
Margie Danchin, paediatrician at the Royal Childrens Hospital and associate professor and clinician scientist, University of Melbourne and MCRI, Murdoch Children’s Research Institute
Archana Koirala, paediatrician and infectious diseases specialist, University of Sydney
Fiona Russell, senior principal research fellow; paediatrician; infectious diseases epidemiologist; vaccinologist, The University of Melbourne
Philip Britton, senior lecturer, child and adolescent health, University of Sydney
This article is republished from The Conversation under a Creative Commons license. Read the original article.


